Hui Zhang, Ying-Jie Wang, Hao-Wen Jiang,* and Peng-Fei Xu*
https://doi.org/10.1021/acs.orglett.5c05168
ABSTRACT: Herein, we report a strategy for the methylalkylation of unactivated alkenes via synergistic photoredox/nickel catalysis. This mild transformation employs di-tert-butyl peroxide (DTBP) as a bifunctional reagent, which serves simultaneously as a hydrogen atom transfer (HAT) reagent and a source of methyl radical, thereby facilitating the coupling of malonate-derived radicals with alkenes. By integrating HAT with radical selectivity control, the method enables the formation of two C(sp 3 ) bonds in a single synthetic operation, offering a streamlined route to valuable three-dimensional scaffolds.
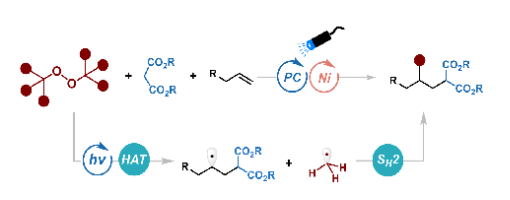
Ai-Lian Wang, Yi-Fan Yao, Xu-Gang Zhang,* and Peng-Fei Xu*
https://doi.org/10.1021/acs.orglett.5c04773
ABSTRACT:
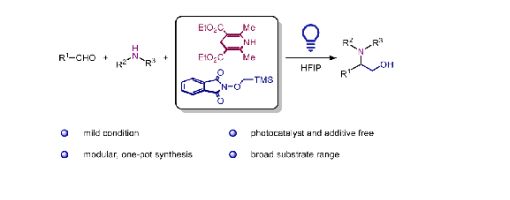
Herein, we report a catalyst-free, photosensitized strategy for synthesizing 1,2-amino alcohols. This transformation proceeds via the addition of an α-hydroxymethyl radical to an in situ-generated alkyliminium ion. The α-hydroxymethyl radical is formed by the radical Brook rearrangement of a (trimethylsilyl)- methoxy radical generated from an electron donor-acceptor (EDA) complex between Hantzsch ester and N-((trimethylsilyl)- methoxy)phthalimide. Therefore, this Mannich-type reaction establishes an additive-free, modular, and simple approach for the synthesis of 1,2-amino alcohols.
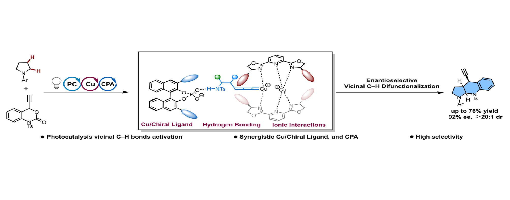
Teng-Fei Xiao, Ke-Rui Jian, Yu-Cheng Gu, Guo-Qiang Xu and Peng-Fei Xu
https://doi.org/10.1039/D5SC06987D
ABSTRACT
Existing strategies are typically limited to modifying a C–H site (a or b-position) of saturated cyclic amines, but the asymmetric difunctionalization of vicinal C–H bonds remains a formidable challenge. To address this challenge, this work introduces a synergistic catalytic system that merges visible-light photocatalysis with asymmetric copper and chiral phosphoric acid catalysis. This system enables the enantioselective synthesis of ring-fused amine skeletons by activating vicinal C–H bonds in straightforward saturated cyclic amines. The reaction proceeds in good yields (up to 76%) and excellent enantioselectivity (up to 92% ee). This work describes detailed mechanistic studies that identify the specific dual chiral catalytic system that forms the basis for the enantioselectivity.